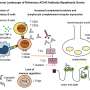
A recent study has identified an immune signature associated with treatment-resistant myasthenia gravis (MG), offering hope for improved patient outcomes. Myasthenia gravis is a rare autoimmune disease where the immune system attacks the connection between nerves and muscles, leading to debilitating muscle weakness. While many patients respond well to existing therapies, a significant subset develops a severe form known as refractory MG, which does not respond to standard treatments.
The research, conducted by a team at the University of California, San Francisco, was published in the Journal of Clinical Investigation in 2023. This study highlights the urgent need for reliable biomarkers that can help clinicians identify which patients will benefit from available therapies and which will not. Currently, the lack of predictive markers complicates treatment plans and can lead to delayed care for those who need it most.
Understanding Myasthenia Gravis
Myasthenia gravis affects various muscle groups, with symptoms that can range from mild to life-threatening. The condition can impair vision, movement, speech, swallowing, and even breathing. Patients with refractory MG often experience a worsening of symptoms despite treatment efforts, which can include medications, plasma exchange, or immunosuppressive therapies.
According to the National Institutes of Health, approximately 20% to 30% of MG patients develop this treatment-resistant form. This highlights the critical need for further research into the underlying mechanisms of the disease and the development of targeted therapies.
The study’s findings suggest that specific immune profiles may be present in patients with refractory MG. By identifying these signatures, healthcare providers may be able to tailor interventions more effectively, potentially improving the quality of life for affected individuals.
Implications for Future Research
The identification of an immune signature is a promising advancement, but researchers emphasize that more work is needed. Future studies could focus on validating these biomarkers in larger patient populations and exploring their potential for guiding treatment decisions.
Moreover, understanding the immune mechanisms at play could lead to the development of innovative therapies that specifically target these pathways. Researchers hope that such advancements will not only enhance treatment for refractory MG but also contribute to a broader understanding of autoimmune diseases.
The findings underscore a significant shift toward personalized medicine in the treatment of myasthenia gravis. With continued research and collaboration within the scientific community, there is optimism that patients facing treatment-resistant forms of MG may find new avenues of hope in the near future.