A research team led by Dr. Sung Mook Choi from the Korea Institute of Materials Science (KIMS) has developed a non-precious metal catalyst that significantly improves the efficiency of hydrogen production through water electrolysis. This new catalyst, designed for anion exchange membrane water electrolysis (AEMWE), offers a promising alternative to traditional precious metal catalysts, which are often expensive and less sustainable.
The innovative catalyst features a layered structure optimized for alkaline conditions, allowing for the use of lower-cost materials without compromising performance. AEMWE has gained attention as a cost-effective and safe method for hydrogen production, but the challenge has been the lack of durable non-precious metal oxygen evolution reaction (OER) catalysts. Many conventional catalysts suffer from degradation and reduced efficiency over time, which has limited their practical application.
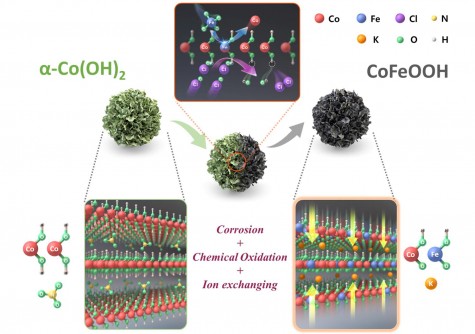
To overcome these limitations, the research team engineered a cobalt (Co) and iron (Fe)-based oxyhydroxide (CoFeOOH) catalyst. This design not only enhances the electronic structure but also improves the reaction pathways during the oxygen evolution process. By incorporating iron into the layered structure, the team could effectively lower the energy barriers associated with critical reaction steps, resulting in high current densities even at low overpotentials.
In addition to enhancing performance, the researchers developed a novel technique for controlling the catalyst’s surface through chemical oxidation. This method stabilizes the catalyst’s structure, preventing corrosion and degradation during operation. The team successfully validated this catalyst in a unit cell of AEMWE, demonstrating its durability and efficiency under real-world electrolysis conditions.
This breakthrough has significant implications for the future of hydrogen production. The new catalyst not only reduces dependence on costly precious metals but also paves the way for the commercialization of high-efficiency AEMWE systems. Once fully developed, this technology could transform the landscape of clean hydrogen production and bolster self-reliance in essential materials for water electrolysis.
Dr. Sung Mook Choi stated, “This research represents a case in which the limitations of non-precious metal–based catalysts were overcome through structural design.” He emphasized the commitment to advancing commercial green hydrogen production technologies that could help realize a hydrogen-based society.
The research received support from the National Research Foundation of Korea (NRF) under the H2 NEXT ROUND program, among other institutional initiatives. The findings were published on December 1, 2025, in the reputable journal ACS Nano, which holds an impact factor of 16.1.
In summary, this advancement in catalyst design marks a significant step towards sustainable and cost-effective hydrogen production, reinforcing the potential for non-precious metal solutions in the energy sector. The ongoing research efforts aim to accelerate the implementation of these technologies, highlighting their role in the future of clean energy.